09 Oct How much do we need? And other protein questions…
In this Thought for Food, we focus on dietary protein. How much do we need? Who needs more and why? When and how often do we need it? How do we assess protein quality? What about sustainability? These were all questions addressed at the 2023 International Symposium on Dietary Protein for Human Health in Utrecht, The Netherlands.
The symposium, co-organised by the FAO, Riddet Institute, and Wageningen University, in cooperation with the International Atomic Energy Agency (IAEA), brought together leading experts from across the globe for three days to discuss the current understanding of this key topic. This provided a great forum to reflect on the advances in our understanding over the 12 years since the FAO Expert Consultation on Dietary Protein Quality Evaluation in Human Nutrition in 2011. Dr Andrew Fletcher presented at the symposium on behalf of the Sustainable Nutrition Initiative® on a global nutrient context of food protein.
The following is a snapshot of some of the key points from the symposium, with references where appropriate to specific speakers.

What is protein and why do we need it?
Protein is not a nutrient. Rather, it is a bundle comprising nine indispensable amino acids (IAAs) and a pool of other (dispensable) amino acids (DAAs) that are all required to support body functions. The IAAs are so named because they must come from our diet as we cannot synthesise them ourselves, whereas the DAAs can be made within the body by transforming other AAs (including any excess IAAs). Dietary protein is shorthand for this collection of amino acids.
Protein is the main structural component of many body tissues – especially muscle – and our view of dietary amino acid requirements has largely been based on the amounts required to support protein turnover and synthesis (Daniel Tomé). However, many amino acids have metabolic roles that are independent of their role as a component in protein synthesis.
An example of this is the IAA leucine: Leucine has importance in muscle protein synthesis (MPS) due to a signalling role, especially among older adults. Elevating the level of leucine in the blood is one of a range of triggers that activate MPS, exercise is another. In the case of a person relegated to bed rest, and unable to exercise, a dietary intervention that temporarily increases leucine may help to offset the muscle wasting that would normally occur. For older and less active adults a doubling of the daily intake target of ~3000 mg may be appropriate (Rajavel Elango).
Other amino acids are critical in immune responses and may be required in greater amounts when combating disease or infection. For instance, the presence of gut parasites increases cystine requirements by 20% (Rajavel Elango) and acute infection may increase IAA requirements to 1.5-2x normal (Mark Manary).
As we improve our understanding of metabolic response to various stresses, we will have more knowledge on how intake targets of various amino acids may be increased to achieve optimal health. However, there is no natural protein with an amino acid composition and content that matches the apparent needs of critical care patients (Mick Deutz).
How often do we need it?
Unlike surplus food energy, or many other nutrients, we do not have a place to store any excess dietary amino acids other than through synthesis of new muscle tissue, which is rate limited. Excess circulating amino acids will gradually be eliminated. This means large single meal intakes will result in excess amino acids being converted to urea and wasted, at the same time placing stress on the liver and kidneys. Rats fed protein-balanced diets across three meals tended to increase their muscle mass, whereas those fed most of the protein in one of the three meals increased their organ mass (Don Layman).
Key factors that impact muscle protein synthesis include exercise, the leucine trigger discussed above, and the overall supply of both IAAs and total AAs. For endurance athletes for example, a post exercise meal containing 0.5 g of quality protein per kg of body mass is probably suitable to optimise muscle remodelling (Daniel Moore).
It is not clear how much time must elapse between meals to “reset” the leucine trigger for muscle protein synthesis. Phil Atherton suggested it needs at least 3-4 hours, and Don Layman reported that many of the signalling compounds are still elevated 5 hours after a first adequate meal, suggesting more time may be required. Most human studies have been conducted on the first meal after an overnight fast. What is clear is that re-feeding too soon does not appear to lengthen or reactivate the process, so snacking to maintain AA levels will not work.
How do we assess protein quality?
The concept of protein quality has been covered in a previous Thought for Food (Protein: we need quality, not just quantity – Sustainable Nutrition Initiative®).
To properly understand the nutritional quality of protein sources, we need information on the absorption of individual amino acids from foods and meals that we typically consume. The way food is prepared may impact the extent of digestion and nutrient absorption. This has been a major focus of work over the last decade, as the FAO Expert Consultation in 2011 concluded there was insufficient data for true ileal AA digestibility in human foods to enable adoption of this approach at the time. Since then, assessments have been made for approximately 500 foods, although these have yet to be brought together into a single database.
A key feature of the digestible amino acid approach is that the protein quality of a meal can be evaluated by summing up the digestible quantities of IAAs from each of the foods. Results combining breakfast cereals and milk indicate that this is the case (Hans Stein). We have used this approach in the DELTA Model® to assess global amino acid supply. This is an improvement on previous methods that just applied a single quality score to the protein, as it can recognise synergies between different types of protein when combined in a meal.
However, measuring amino acid digestibility is not simple. We cannot just measure the difference between amino acids entering as food and leaving via faeces, as a significant portion (up to 80%) of faecal amino acid content is from a combination of lower intestinal secretions and bacterial growth (Suzanne Hodgkinson). Instead, we need to measure the difference between intake and the amount present at the end of the small intestine (ileum). Direct measurement requires either trials with human ileostomates (often recovering bowel cancer patients; Suzanne Hodgkinson), or specially canulated pigs (which have a very similar digestive system; Hans Stein). Both methods are challenging and expensive and there has been a lot of work directed at finding alternatives. Two approaches have emerged:
- A dual isotope method that uses stable isotopes of carbon (13C) and hydrogen (2H) to track absorption of labelled amino acids. This is one reason why the IAEA was involved with the symposium. This method can be used with a wide range of subjects to better understand the impact of physiological state on amino acid absorption and utilisation. However, we need to be able to produce foods containing the isotope labelled amino acids.
- In vitro systems that simulate human digestive processes, including the mechanical mixing actions that occur in the stomach. Once calibrated against in vivo data these systems provide a means to test a wide range of foods. Session three of the symposium was dedicated to discussion of these systems.
Digestible Indispensable Amino Acid Score (DIAAS)
At times it is useful to have a single metric that conveys the comparative quality of a protein source. Historically this has been the Protein Digestibility Corrected Amino Acid Score (PDCAAS). The replacement for this is DIAAS, which reflects the delivery of the most limiting amino acid relative to its requirement within a food or meal. However, the real advance over the last decade is not changing from PDCAAS to DIAAS but shifting to the more complete digestible IAA approach that can be additive across meal intakes and dietary patterns to best assess if specific AA requirements are met. This cannot be achieved with DIAAS values but requires the underlying data instead (Paul Moughan).
Currently there is not a consistent approach to protein quality in food labelling regulations, and the information conveyed to consumers is limited. In many cases protein quality is not a factor, only protein quantity. Where protein quality does factor into labelling requirements (e.g., USA) this is fixed in law, and may be difficult to change to reflect the latest science.
Sustainability
Food system sustainability starts with providing adequate nutrition. Achieving the 2030 sustainable development goal of Zero Hunger remains a challenge for the global community, especially for many countries in Sub-Saharan Africa (Barbara Burlingame). Protein has a role in this, but it is not the whole story.
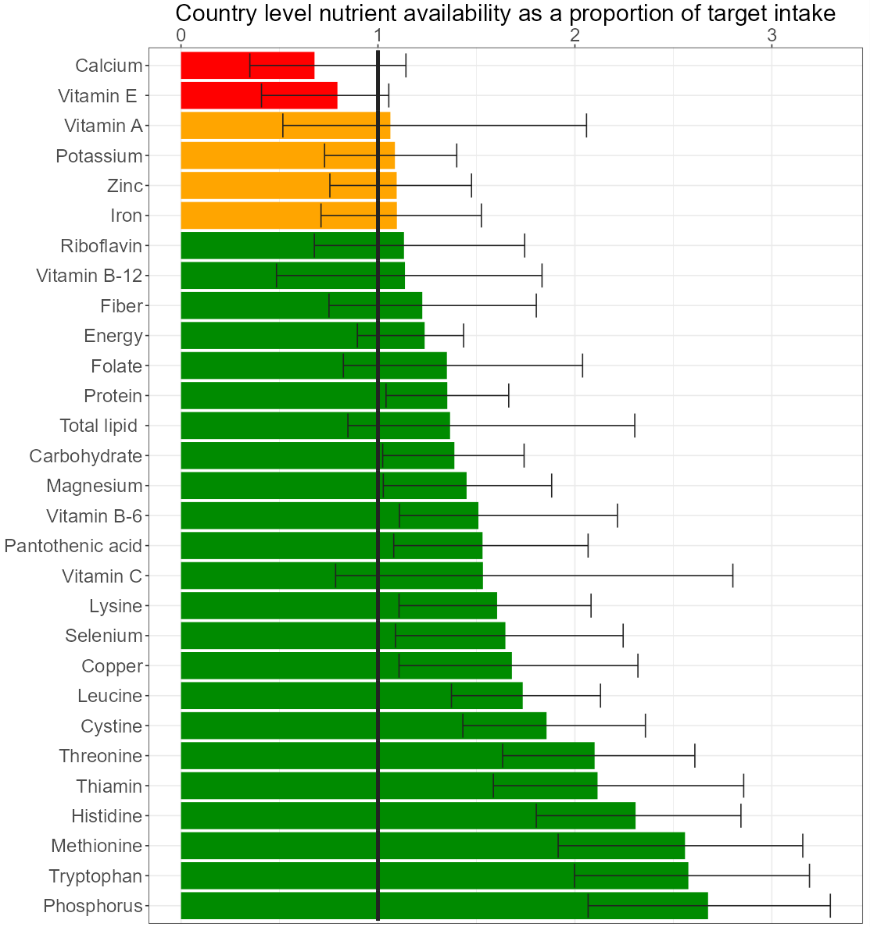
Findings from our work with the DELTA Model® (Andrew Fletcher) show that neither protein nor the IAA lysine is a limiting nutrient globally but may be undersupplied for some countries (Figure 1).
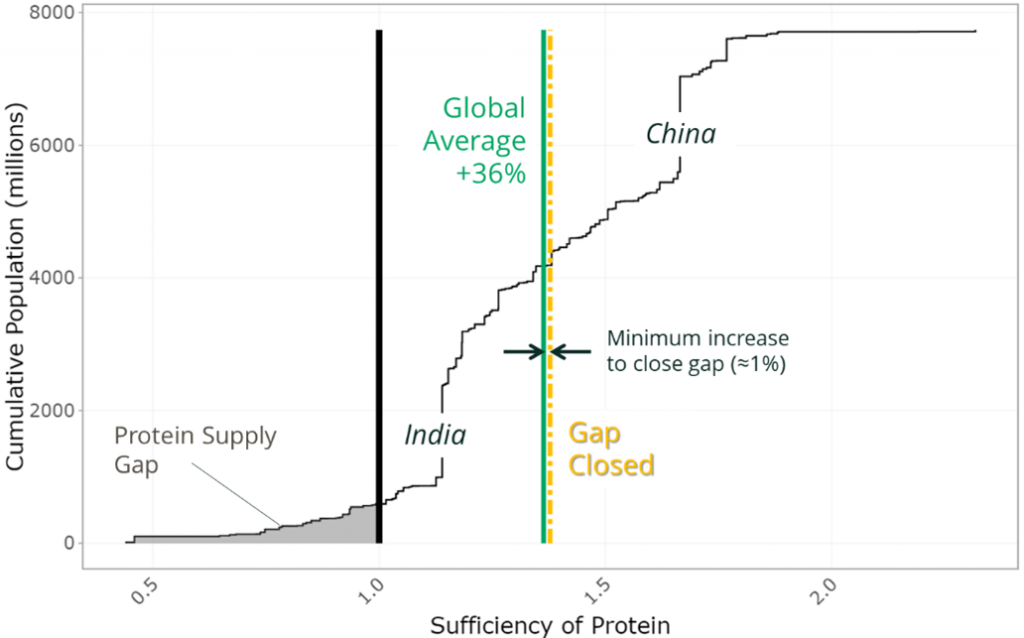
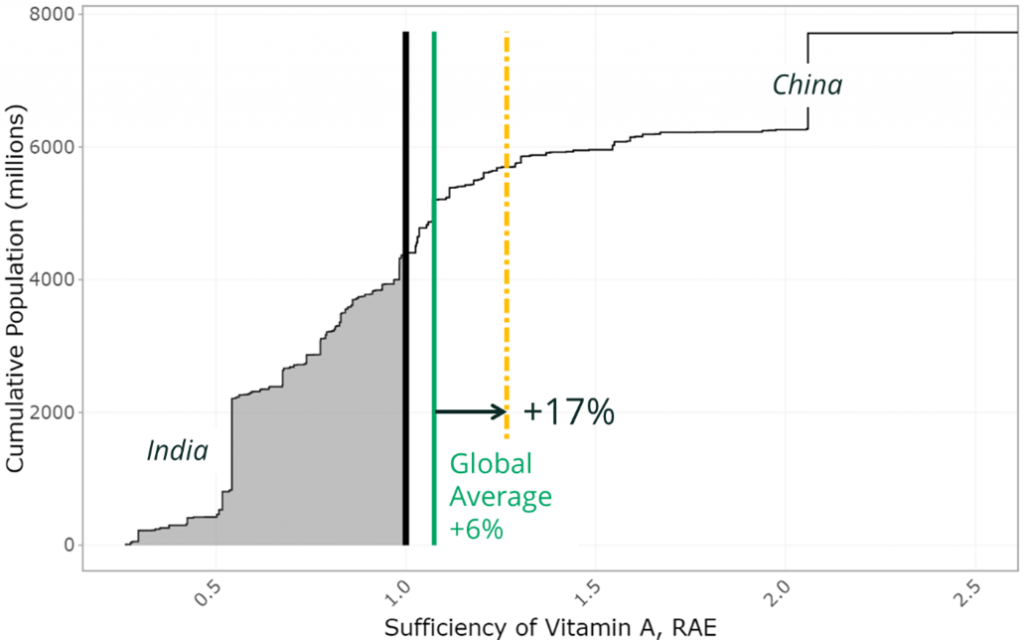
Micronutrients such as calcium, riboflavin and vitamin A have a greater gap for many countries. Thus, a larger increase is needed from our food supply of these nutrients to meet requirements. For example, to close the 2020 protein gap for countries that were deficient would have required a 1% increase in global protein supply, whereas a 17% increase in vitamin A would have been required to address country level supply gaps of this nutrient (Figure 2).
Rather than focus on protein alone it is more important to consider the role that protein-rich foods might play in closing other nutrient gaps.
Animal and plant-sourced foods provide complementary sources of nutrients to a diet. Both plant (dark leafy green vegetables) and animal-sourced foods such as eggs and dairy are sources of commonly lacking nutrients and are often found in the least-cost or affordable diets (Sylvia Chungchunlam). Provision of these nutrient-dense foods remains a priority for low- and middle-income countries, especially for populations with higher needs.
When we look at the novel protein production methods (fermentation, single cell protein) there is large variation in the environmental impacts associated with these and the nature of the electricity supply plays a key role in emissions (Hanna Tuomisto). Many of these technologies are at a comparatively early stage of development which brings additional uncertainty on the performance that can be realised. We also need to consider that many of these products are ingredients rather than finished foods.
In a “circular food system”, there are combinations of plant and animal production that make the best use of environmental resources in a region to deliver nutrition (Hannah Van Zanten). The specific systems are highly dependent on the food trade conditions set at the boundary. For European models, a nutritional self-sufficiency objective gives a very different result from an objective to provide nutrition to other countries. However, all scenarios involve a combination of plants and animals.
To conclude
This is a sample of the ideas discussed. All these aspects are important as we consider how the global food system might evolve to meet the nutritional needs of the expanding global population while respecting environmental constraints. There are still unanswered questions, but we are in a much better position than we were 12 years ago. The more we understand what we really need, and how different foods contribute, the better we can create credible options for the future.
This Thought for Food was written by Dr Andrew Fletcher, and Patricia Soh, with the support of the SNi team.